The next step to increasing the power of the room-temperature battery comes in improving the electrolytes - the components that allow the electrical charge to flow through the battery.įor more information, contact Nat Levy at This email address is being protected from spambots. The liquid-metal battery’s lower cost arises from simpler materials, chemistry, and system design compared to lithium-ion, and its longer lifetime, says Sadoway. Finding alternative materials that can deliver the same performance while reducing the cost of production remains a key challenge. A liquid metal battery comprises two liquid metal electrodes separated by a molten salt electrolyte that self-segregate into three layers based upon density and. One representative group is the family of rechargeable liquid metal batteries, which were initially exploited with a view to implementing intermittent energy sources due to their specific benefits including their ultrafast electrode charge-transfer kinetics and their ability to resist microstructural electrode degradation. Many of the elements that constitute the backbone of the new battery are more abundant than some of the key materials in traditional batteries, making them potentially easier and less expensive to produce on a large scale however, gallium remains an expensive material. 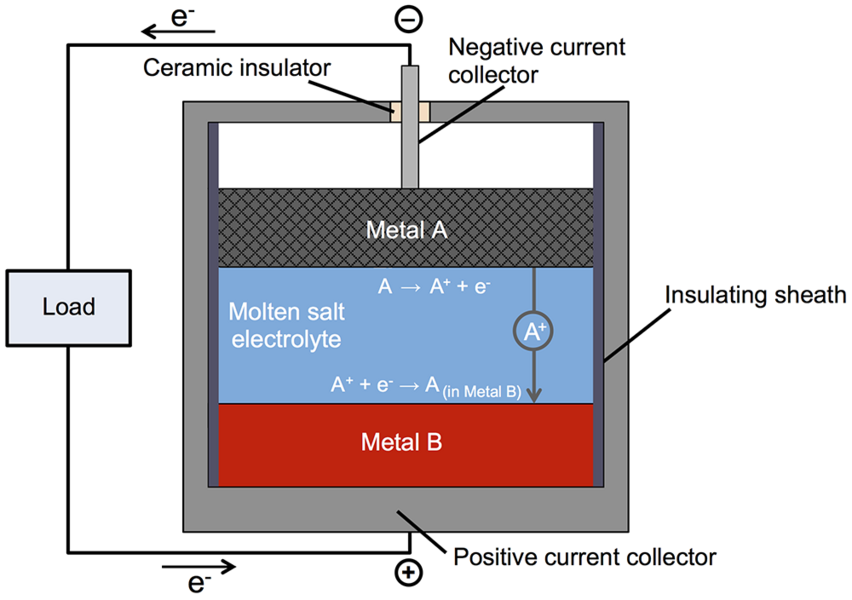
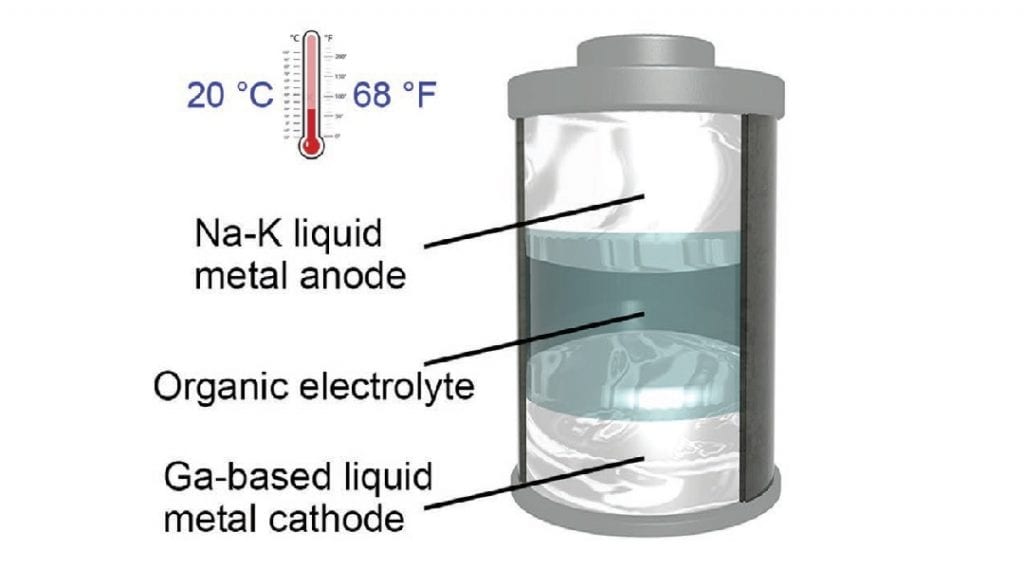
That flexibility allows these batteries to potentially power everything from smartphones and wearable devices to the infrastructure underpinning the movement toward renewable energy. It may be possible to create a battery with even lower melting points using different materials.īecause of the liquid components, the battery can be scaled up or down easily, depending on the power needed -the bigger the battery, the more power it can deliver. The battery includes a sodium-potassium alloy as the anode and a gallium-based alloy as the cathode. The metallic electrodes in the new battery can remain liquefied at a temperature of 20 ☌ (68 ☏) - a major change because current liquid-metal batteries must be kept at temperatures above 240 ☌. Liquid-state batteries can deliver energy more efficiently without the long-term decay of sold-state devices however, they either fall short on high energy demands or require significant resources to constantly heat the electrodes and keep them molten. Solid-state batteries feature significant capacity for energy storage but they typically encounter numerous problems that cause them to degrade over time and become less efficient. The researchers have created a “room-temperature all-liquid-metal battery,” which includes the best of both worlds of liquid-and solid-state batteries. Most batteries are composed of either solid-state electrodes, such as lithium-ion batteries for portable electronics, or liquid-state electrodes including those for smart grids. Researchers have built a new type of battery that combines the benefits of existing options while eliminating their key shortcomings and saving energy. University of Texas, Austin, TX The battery includes a sodium-potassium alloy as the anode and a gallium-based alloy as the cathode. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
